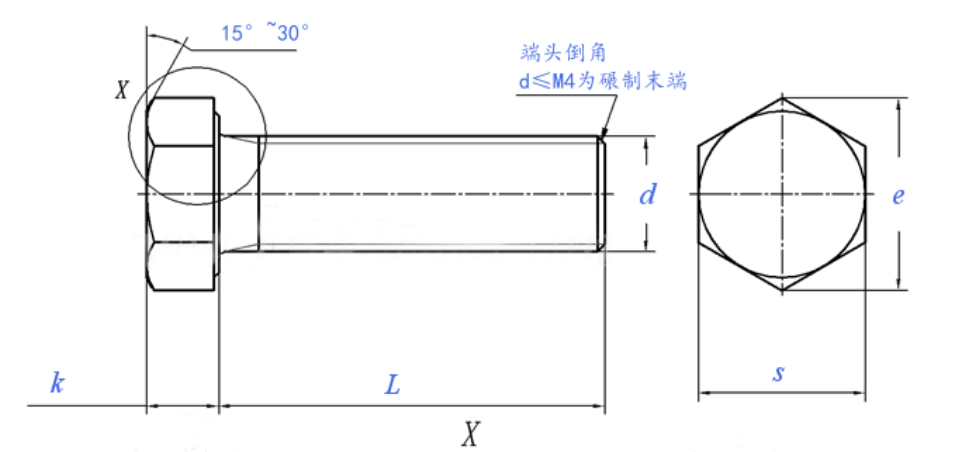
Introduction
Zinc plated hex bolts are externally threaded fasteners commonly used in a wide array of industrial applications, including automotive, construction, and machinery. They consist of a hexagonal head and a threaded shank, manufactured from carbon or alloy steel and subsequently coated with a layer of zinc. This zinc coating serves as a crucial barrier against corrosion, significantly extending the service life of the fastener in diverse environmental conditions. Their widespread adoption is due to a balance of strength, cost-effectiveness, and corrosion resistance, making them a staple in many bolted joint designs. The technical position of these bolts is central to mechanical fastening, functioning as a key component in ensuring structural integrity and load distribution. Core performance characteristics include tensile strength, yield strength, torque retention, and resistance to hydrogen embrittlement – all critical factors in ensuring reliable and safe operation.
Material Science & Manufacturing
The base material for zinc plated hex bolts is typically medium carbon steel, such as ASTM A307 Grade A or equivalent, chosen for its balance of strength and ductility. Alloy steels, such as ASTM A307 Grade B, may be used for applications requiring higher strength. The steel undergoes a cold forming process – generally upset forging – to create the hex head shape, increasing the grain flow along the fastener's length and improving strength. The threading is then produced via rolling, which work-hardens the steel and provides a smooth, accurate thread profile. Crucially, the steel’s microstructure is characterized by pearlite and ferrite phases; the proportion of each dictates the material’s hardness and toughness.
Zinc plating is typically achieved via hot-dip galvanizing or electroplating. Hot-dip galvanizing provides a thicker, more durable coating, but can result in a less uniform finish. Electroplating offers tighter control over coating thickness and surface finish. Prior to plating, the steel is meticulously cleaned – degreased, pickled (to remove scale), and rinsed – to ensure proper zinc adhesion. The zinc coating itself is a layered structure, consisting of a zinc-iron alloy layer closest to the steel substrate, followed by layers of pure zinc. The thickness of the zinc coating is a critical parameter, typically ranging from 5 to 25 micrometers depending on the desired level of corrosion protection. Parameters like bath temperature, current density (in electroplating), and steel surface preparation are meticulously controlled to achieve consistent coating quality and prevent defects such as porosity or blistering. Hydrogen embrittlement, a common issue during electroplating, is mitigated through post-plating baking processes.

Performance & Engineering
The performance of zinc plated hex bolts is governed by several key engineering principles. Tensile strength, the maximum stress the bolt can withstand before fracture, is directly related to the steel grade and diameter. Yield strength, the stress at which permanent deformation begins, is a critical factor in preventing loosening under load. Torque-tension relationships are carefully considered during joint design; the applied torque must correlate to the desired preload, ensuring adequate clamping force.
Environmental resistance is paramount. The zinc coating provides galvanic protection to the steel substrate; zinc corrodes preferentially, sacrificing itself to protect the underlying steel. However, this protection is limited by the coating thickness and the presence of discontinuities. In aggressive environments (e.g., saltwater, acidic atmospheres), the zinc coating will eventually deplete, leading to corrosion of the steel. Furthermore, chloride ions can penetrate the zinc coating, accelerating corrosion. The rate of corrosion is also influenced by temperature and humidity. Compliance requirements, such as RoHS (Restriction of Hazardous Substances) and REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals), dictate restrictions on the use of certain chemicals in the plating process and the allowable levels of hazardous substances in the finished product. The design engineer must account for these constraints when selecting zinc plating processes.
Fatigue loading, common in dynamic applications, presents a significant challenge. Cyclic stresses can lead to crack initiation and propagation, ultimately resulting in bolt failure. The surface finish of the bolt and the quality of the zinc coating play a crucial role in fatigue performance; surface defects can act as stress concentrators, accelerating crack growth. Preload is also a critical factor; higher preload levels can improve fatigue resistance by reducing stress range.
Technical Specifications
| Diameter (mm) | Material Grade | Zinc Coating Thickness (µm) | Tensile Strength (MPa) |
|---|---|---|---|
| M6 | ASTM A307 Grade A | 8-12 | 400-550 |
| M8 | ASTM A307 Grade A | 8-12 | 400-550 |
| M10 | ASTM A307 Grade B | 15-25 | 600-750 |
| M12 | ASTM A307 Grade B | 15-25 | 600-750 |
| M16 | SAE J429 Grade 5 | 20-30 | 830-1040 |
| M20 | SAE J429 Grade 8 | 25-35 | 950-1200 |
Failure Mode & Maintenance
Zinc plated hex bolts are susceptible to several failure modes. Corrosion is a primary concern, particularly in harsh environments. Crevice corrosion can occur under washers or other contact points, accelerating localized corrosion. Hydrogen embrittlement, induced during electroplating, can lead to delayed cracking, particularly under tensile stress. This is exacerbated by high strength steels. Fatigue failure, as previously mentioned, occurs due to cyclic loading. Stripping of threads is another common failure mode, resulting from excessive torque or improper thread engagement. Shear failure can occur if the bolt is subjected to excessive shear loads.
Maintenance procedures are crucial for preventing premature failure. Regular inspection for signs of corrosion is essential. Applying a corrosion inhibitor or protective coating can extend the service life of the bolts. Proper torque control during installation and re-tightening is critical to prevent stripping and ensure adequate clamping force. Lubrication of threads can reduce friction and improve torque accuracy. If hydrogen embrittlement is a concern, baking the bolts after plating is recommended. In cases of severe corrosion or damage, the bolts should be replaced immediately. Periodic re-tightening, based on application-specific requirements and environmental conditions, is also recommended. Consideration should be given to replacing zinc plated bolts with stainless steel or other corrosion-resistant materials in particularly aggressive environments.
Industry FAQ
Q: What is the difference between hot-dip galvanizing and electroplating for zinc coating, and which is preferred for high-stress applications?
A: Hot-dip galvanizing provides a thicker zinc coating (typically 50-150µm) with excellent corrosion resistance due to the formation of a zinc-iron alloy layer. However, the process can result in a rougher surface finish and potential distortion of the bolt. Electroplating offers a thinner, more uniform coating (typically 5-25µm) with a smoother surface finish, but provides less corrosion protection. For high-stress applications, the smoother surface finish of electroplating is often preferred to minimize stress concentrations and improve fatigue resistance, despite the lower corrosion protection. Supplementary corrosion protection methods, like post-treatment coatings, can be used with electroplated bolts for increased durability.
Q: How does the zinc coating affect the torque-tension relationship of a hex bolt?
A: The zinc coating slightly increases the effective diameter of the bolt threads, which can lead to a minor reduction in preload for a given applied torque. This effect is typically accounted for in joint design by adjusting the torque specification or using lubricated fasteners. The thickness and uniformity of the zinc coating are critical factors; inconsistent coating can introduce variations in friction and preload.
Q: What measures can be taken to mitigate hydrogen embrittlement in zinc plated high-strength bolts?
A: Hydrogen embrittlement is a significant concern with high-strength steels. Mitigation strategies include baking the bolts after plating to drive out absorbed hydrogen, using a plating process that minimizes hydrogen absorption, and applying a post-treatment coating to act as a diffusion barrier. Selecting a lower strength steel grade can also reduce susceptibility to hydrogen embrittlement, but may compromise the overall strength of the joint.
Q: What are the limitations of zinc plating in saltwater environments?
A: Zinc plating provides limited protection in saltwater environments. Chloride ions accelerate the corrosion of zinc and, once depleted, the underlying steel. The rate of corrosion is significantly higher in saltwater than in freshwater or atmospheric conditions. In such environments, stainless steel bolts, or bolts with more robust corrosion protection (e.g., cadmium plating, although environmentally restricted, or specialized coatings like zinc-nickel), are recommended.
Q: How do different zinc alloy coatings (e.g., zinc-nickel) compare to standard zinc plating in terms of corrosion resistance?
A: Zinc-nickel alloy coatings offer significantly superior corrosion resistance compared to standard zinc plating. They provide a more stable and protective layer, even in harsh environments. Zinc-nickel coatings are particularly effective in resisting white rust (zinc corrosion products) and red rust (iron oxide). However, they are typically more expensive than standard zinc plating and may require specialized application processes.
Conclusion
Zinc plated hex bolts remain a critical fastening solution across numerous industries due to their cost-effectiveness and ability to provide adequate corrosion protection in many applications. The performance of these fasteners is intricately linked to the underlying material science of the steel substrate, the precision of the manufacturing processes, and the quality control measures implemented during zinc plating. Understanding the potential failure modes – corrosion, hydrogen embrittlement, and fatigue – is crucial for ensuring long-term reliability and safety.
Future advancements in zinc plating technologies, such as the development of more environmentally friendly plating processes and the application of nano-coatings, promise to further enhance the performance and sustainability of zinc plated hex bolts. Careful consideration of environmental factors, load requirements, and applicable industry standards remains paramount when selecting and utilizing these fasteners. Continued research and development in corrosion protection and materials science will be essential for extending the lifespan and improving the performance of these ubiquitous components.

